|
However, it's easy to determine the configuration of electrons for heavier elements by making a chart. If there are more electrons than protons, the ion has a negative charge and is called an anion.Įlements are shown from atomic number 1 (hydrogen) up to 94 (plutonium). If there are more protons than electrons, an atomic ion has a positive charge and is called a cation. Atomic mass of Nitrogen element is 14.007 u, Atomic mass of Oxygen element is 15.999 u. Let me tell you the atomic mass of Nitrogen and oxygen. I have explained the concept of valence electrons for d block and f block elements in separate articles d block elements. Antimony has many isotopes, but out of them the most abundant isotope is 121 Sb (around 57). The crystal structure of antimony is Rhombohedral. The melting point of antimony is 630.6 ☌ and its boiling point is 1635 ☌. The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons-or not.Īn ion of an atom is one in which the number of protons and electrons is not the same. electrons, the VIIA family has 7valence electrons, and the VIIIA family has 8 valence electrons. However, this value is very small and the reason behind such a small value is the difference in atomic mass of Nitrogen and oxygen. Youll get labeled Periodic table with Name, Atomic mass, Electron configuration, Electronegativity, Ionization energy, Electron affinity etc. The atomic mass of antimony is 121.76 u and its density is 6.7 g/cm 3. Remember, a neutral atom contains the same number of protons and electrons. The upper right side shows the number of electrons in a neutral atom. The easiest method to keep in mind electron layouts is usually to content label the Routine Dinner table in blocks of factors. The elements in same Periods have the same number of energy shells (or energy orbits). The names of the periods in periodic table are Period 1: Shortest period. The element atomic number and name are listed in the upper left. There are total 7 periods on the periodic table. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. The electron shells are shown, moving outward from the nucleus. As described in Section 10.6, the modern periodic table is arranged based on an atoms valence electrons. Here are electron shell atom diagrams for the elements, ordered by increasing atomic number.įor each electron shell atom diagram, the element symbol is listed in the nucleus. For that, we have electron shell diagrams. The periodic table, also known as the periodic table of the elements, is an ordered arrangement of the chemical elements into rows ('periods') and columns ('groups'). Hence the phosphorus element has electrons arrangement 2, 8, 5. 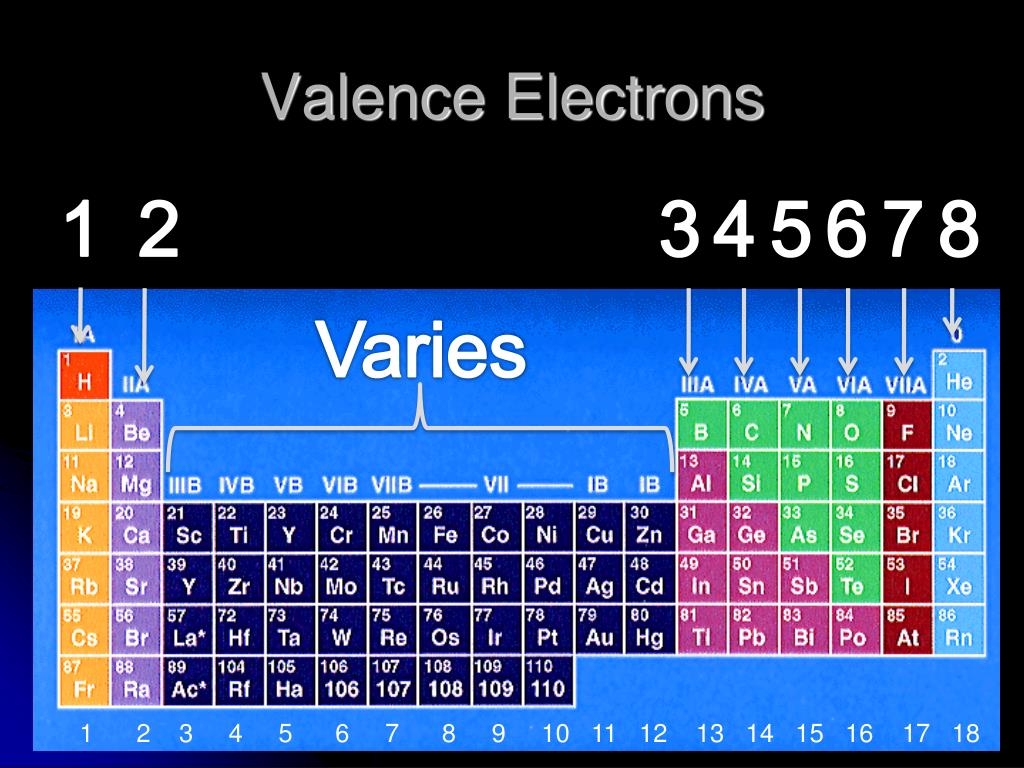 
It's easier to understand electron configuration and valence if you can actually see the electrons surrounding atoms. Now the atomic number of phosphorus (P) is 15.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
